Sunday, June 30, 2013 at 10:29AM
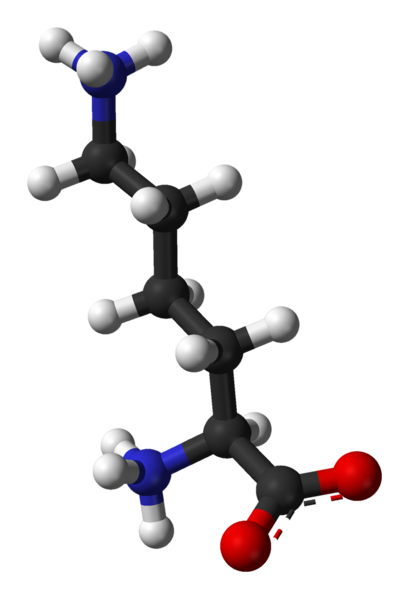
Sunday, June 30, 2013 at 10:29AM "Ethanolamine, also called 2-aminoethanol or monoethanolamine (often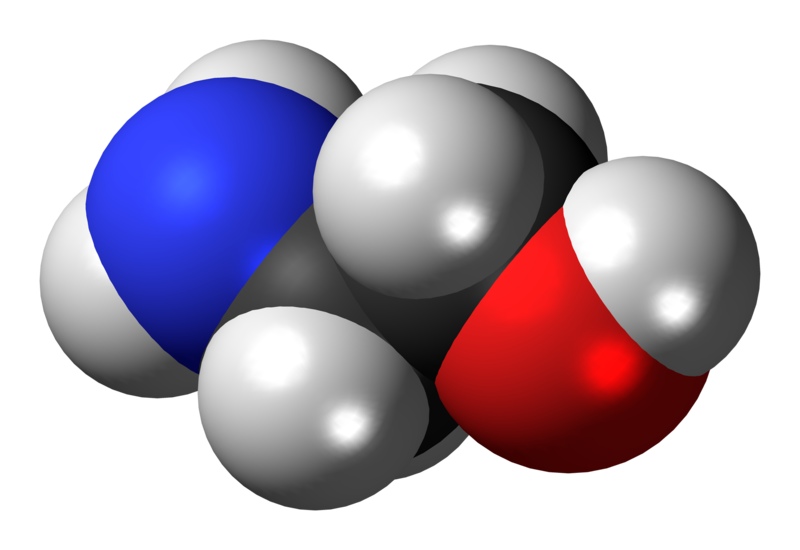 abbreviated as ETA or MEA), is an organic chemical compound that is both a primary amine and a primary alcohol (due to a hydroxyl group). Like other amines, monoethanolamine acts as a weak base. Ethanolamine is a toxic, flammable, corrosive, colorless, viscous liquid with an odor similar to that of ammonia."
abbreviated as ETA or MEA), is an organic chemical compound that is both a primary amine and a primary alcohol (due to a hydroxyl group). Like other amines, monoethanolamine acts as a weak base. Ethanolamine is a toxic, flammable, corrosive, colorless, viscous liquid with an odor similar to that of ammonia."
"Ethanolamine is commonly called monoethanolamine or MEA in order to be distinguished from diethanolamine (DEA) and triethanolamine (TEA). Ethanolamine is the second-most-abundant head group forphospholipids, substances found in biological membranes, and is also used in messenger molecules such as palmitoylethanolamide which has an effect on CB1 receptors."
"The term ethanolamines (plural) is a group of amino alcohols. A class of antihistamines is identified as ethanolamines which includes carbinoxamine, clemastine, dimenhydrinate, diphenhydramine, and doxylamine."